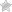The informed consent form for research is a document that ensures that a participant in a medical research project have been fully educated on the nature of the project and have been given ample opportunity to ask any questions they need to fill comfortable and informed. The form contains a list of statements which must be checked off before the document can be signed to indicate to participants full consent.
How to Write
Step 1 – Download in Adobe PDF (.pdf) or Microsoft Word (.docx).

Informed Consent Form – Adobe PDF – Microsoft Word (.docx)
Step 2 – Enter your name in the topmost field. Below this, in the field presented in the first statement, enter the date indicated on the information sheet. Check the first box if you have read and understood the information pertaining to the project. Check the second to show that you’ve had the opportunity to ask questions. Check the third to volunteer to participate.
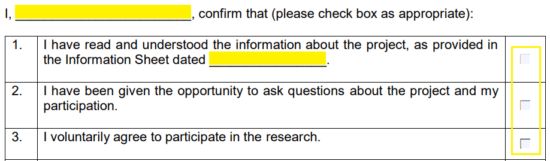
Step 3 – Window 4 states that you can withdraw from the project at any point, for any reason, and you will not be penalized nor will you be asked to provide your reasoning. The fifth statement has to do with confidentiality and checking the corresponding box indicates that the confidentiality agreement has been clearly stated. If there are separate terms of consent for interviews and the like, those terms must be explained to you. If they have, check the box in window six (6).

Step 4 – The use of the data must be explained. If it has, check the applicable box. Other researchers will be privy to the data collected during the project and if they agree to the confidentiality terms, they will be given access. Check the box to approve this statement. You may select whether or not you would like your name used in the ninth window. The final window, simply pertains to the signing and dating of the form.

Step 5 – Print your name and the date where applicable before signing the document and given the researchers your consent to share the information as they deem necessary.